Product:Hydantoin
CAS NO:461-72-3
Synonyms:glycolylurea;2,4-IMIDAZOLIDINEDIONE;2,4-IMIDAZOLINEDIONE;2,4-(3H,5H)-IMIDAZOLEDIONE;Imidazolidine-2,4-dione;
Hydantoin 461-72-3 Package:25 kg bag or drum
Hydantoin 461-72-3 Structure:
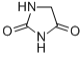
Hydantoin 461-72-3 Specs:
Appearance:White crystals
Loss on drying:0.5%max
Iron(As Fe):0.002%max
Heavy metals(As Pb):0.002%max
Assay:98.5%max
Sulphate:0.3%max
Hydantoin 461-72-3 Application:
Hydantoin, or glycolylurea, is a heterocyclic organic compound with the formula CH2C(O)NHC(O)NH. It is a colorless solid that arises from the reaction of glycolic acid and urea. It is an oxidized derivative of imidazolidine. In a more general sense, hydantoins can refer to a groups and a class of compounds with the same ring structure as the parent. For example, phenytoin (mentioned below) has two phenyl groups substituted onto the number 5 carbon in a hydantoin molecule.
History of Hydantoin 461-72-3:
Hydantoin was first isolated in 1861 by Adolf von Baeyer in the course of his study of uric acid. He obtained it by hydrogenation of allantoin, hence the name.
Urech in 1873 synthesized 5-methylhydantoin from alanine sulfate and potassium cyanate in what is now known as the Urech hydantoin synthesis.The method is very similar to the modern route using alkyl and arylcyanates. The 5,5-dimethyl compound can also be obtained from acetone cyanohydrin (also discovered by Urech: see cyanohydrin reaction) and ammonium carbonate.
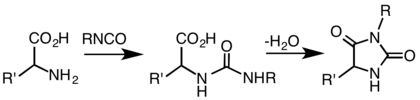
Of practical importance, hydantoins are obtained by condensation of a cyanohydrin with ammonium carbonate. Another useful route, which follows the work of Urech, involves the condensation of amino acids with cyanates and isocyanates:
Related Products:139-07-1,CAS 60-12-8,507-20-0,90-64-2,heptane 142-82-5,25895-60-7,420-04-2,156-62-7.