Product:DL-Mandelic acid 90-64-2
CAS NO:90-64-2
90-64-2 Synonyms:acidomandelico;Almond acid;alpha-hydroxy-alpha-toluicaci;alpha-hydroxy-benzeneaceticaci;alpha-Phenylhydroxyacetic acid;alpha-Toluic acid, alpha-hydroxy-;Amigdalinicacid;Amygdalinic acid
90-64-2 Package:25 kg drum
90-64-2 Structure:
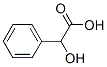
Mandelic acid 90-64-2 Specs:(Cosmetic Grade)
Appearance:White crystals
Assay:99%min
Solubility:soluble in water and ether
MP:118-121
Assay(Cl-):0.01%max
Heavy Metal:20ug/g max
Residue on ignition:0.1%max
Loss on drying:0.5%max
Mandelic acid 90-64-2 Application:
Mandelic acid has a long history of use in the medical community as an antibacterial, particularly in the treatment of urinary tract infections.It has also been used as an oral antibiotic, and as a component of "chemical face peels", along with other alpha-hydroxy acids (AHAs).
1.The drugs cyclandelate and homatropine are esters of mandelic acid.
2.Pemoline is synthesized by the condensation of the ethyl ester of mandelic acid with guanidine.
3.Amolanone
Origin of Mandelic acid 90-64-2:
Mandelic acid was discovered in 1831 by the German pharmacist Ferdinand Ludwig Winckler (1801–1868) while heating amygdalin, an extract of bitter almonds, with diluted hydrochloric acid.The name is derived from the German "Mandel" for "almond". Derivatives of mandelic acid are formed as a result of metabolism of adrenaline and noradrenaline by monoamine oxidase and catechol-O-methyl transferase.
Synthesis of Mandelic acid 90-64-2:
Mandelic acid is usually prepared by the acid-catalysed hydrolysis of mandelonitrile,which is the cyanohydrin of benzaldehyde.Mandelonitrile can also be prepared by reacting benzaldehyde with sodium bisulfite to give the corresponding adduct,forming mandelonitrile with sodium cyanide, which is hydrolyzed:

Related Products:139-07-1,CAS 60-12-8,507-20-0,90-64-2,heptane 142-82-5,25895-60-7,420-04-2,156-62-7.